
J. R. Smyth, C. M. Holl,
H. M. S. Laustsen
Department of Geological Sciences, University of Colorado,
Boulder, CO 80309, USA
F. Langenhorst, C. A. McCammon
Bayerisches Geoinstitut, Universität Bayreuth,
D-95440 Bayreuth, Germany
G. R. Rossman
Division of Geological and Planetary Sciences, California
Institute of Technology, Pasadena, CA 91125, USA
A. Kleppe
Department of Earth Sciences, University of Oxford, Parks Road,
Oxford, OX1 3PR, UK
T. Kawamoto
Institute for Geothermal Sciences, Kyoto University,
874-0903 Beppu, Japan
P. A. van Aken
Institut für Angewandte Geowissenschaften, Technische
Universität Darmstadt, D-64287 Darmstadt, Germany
Wadsleyite II is a variably hydrous magnesium-iron silicate phase similar to spinelloid IV and a potential host for H in the Transition Zone of the Earth's mantle. Two separate samples of wadsleyite II synthesized at 17.5 GPa and 1400°C and at 18 GPa and 1350°C have been characterized by electron microprobe, single-crystal X-ray diffraction, visible, IR, Raman, and Mössbauer spectroscopies, and transmission electron microscopy including electron energy-loss spectroscopy. The two samples have the following chemical formulae: Mg1.71Fe0.18Al0.01H0.33 Si0.96O4 and Mg1.60Fe0.22Al0.01 H0.44Si0.97O4. Mössbauer spectroscopy and electron energy loss spectroscopy (EELS) indicate that about half of the iron present is ferric. Refinement of the structures shows them to be essentially the same as spinelloid IV. Calculated X-ray powder diffraction patterns show only subtle differences between wadsleyite and wadsleyite II. The hydration mechanism appears to be protonation of the non-silicate oxygen (O2) and possibly the oxygens surrounding the partially vacant tetrahedral site Si2, charge-balanced by cation vacancies in Si2, M5 and M6. The unit cell volume of this phase and its synthesis conditions indicate that it may be an intermediate phase occurring between the fields of wadsleyite and ringwoodite, if sufficient trivalent cations are available. The unit cell parameters have been refined at pressures up to 10.6 GPa by single-crystal X-ray diffraction in the diamond anvil cell. The refined bulk modulus for the sample containing 2.8 wt% H2O is 145.6 ± 2.8 GPa with a K' of 6.1 ± 0.7. Similar to wadsleyite and ringwoodite, hydration has a large effect on the bulk modulus. The presence of this phase in the mantle could serve to obscure the seismic expression of the phase boundary between wadsleyite and ringwoodite near 525 km. The large apparent effect of hydration on bulk modulus is consistent with hydration having a larger effect on seismic velocities than temperature in the Transition Zone.

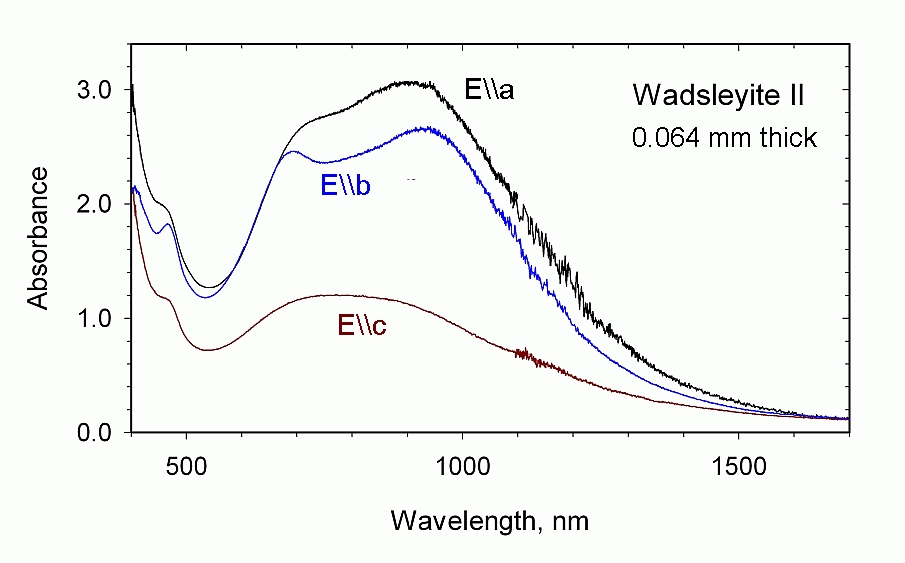
The optical spectrum of wadsleyite II
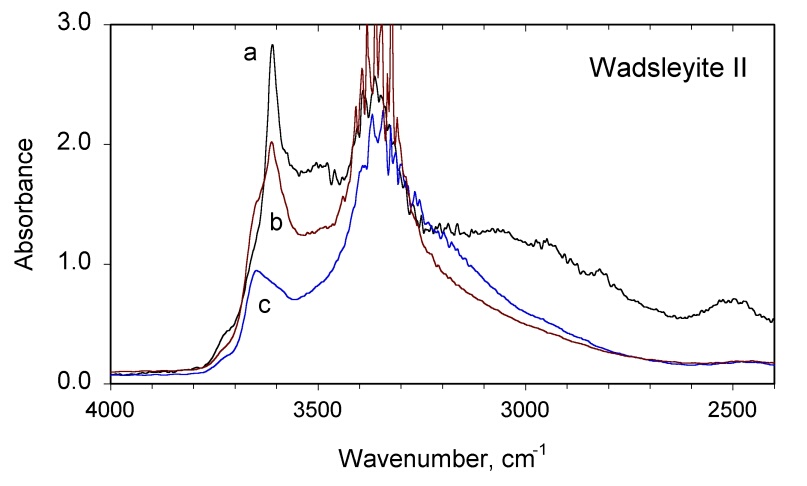
The infrared spectrum in the OH region