Mineralogisches Institut, Universität Heidelberg, D-69120 Heidelberg, Germany.
Abstract
Chemical, structural, infrared, optical
and Mössbauer spectroscopic data were obtained on tourmalines from gem pockets
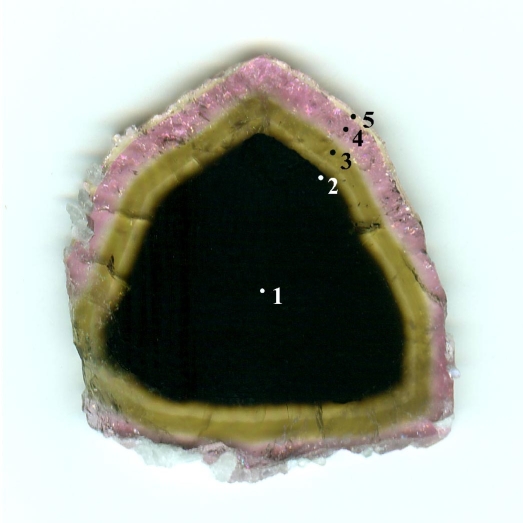
in the Himalaya mine, San Diego County, California, U.S.A., including a strongly
color zoned crystal. Ca
and Li abundances increase from core to rim whereas Mn2+ and F others
increase, peak, and decrease. When lepidolite begins crystallizing, the F
content in tourmaline decreases. The black core of this crystal is a Mn-bearing
“oxy-schorl”. A yellowish green, intermediate Mn-rich “fluor-elbaite” zone
contains a relatively high Mn content with ~6 wt% MnO. The nearly colorless
“fluor-elbaite” rim has the highest Li content of all zones. There is an
inverse correlation between the lattice
parameter a (for values ³ 15.84 Å) and the Li content (r2 = 0.96). Mössbauer studies from the different zones
within this crystal show that the
relative fraction of Fe3+ increases continuously from the Fe-rich
core to the Fe-poor near-rim zone, reflecting the increasing fugacity of oxygen
in the pegmatite pocket. There
is a high positive correlation between the lattice parameter a (for values ³ 15.84 Å) and the (Fe2+ + Mn2+) content in
tourmalines from the elbaite-schorl series (r2 = 0.99). Lower values than 15.84 Å for
a can be suggested to result from an increasing [4]B
content in samples that usually have a (Fe2+ + Mn2+)
content of < 0.1 apfu.
Positive correlations between Al at the Y site and [4]B (r2
= 0.93), and between (Mn2+ + Fe2+) and [4]Al (r2 = 0.99) were found in tourmalines from the Himalaya Mine. These
correlations indicate that, in the short-range order configurations, YAl is coupled with [4]B,
whereas Mn2+ and Fe2+ are coupled with [4]Al.
To
obtain the most accurate OH data for the investigated tourmaline samples the OH
determinations in this study were undertaken by different methods (SIMS,
hydrogen manometry, continuous-flow mass spectrometry). Our analyses indicate that
some elbaites contain a mixed occupation of F, OH and O at the W site. We conclude that the approximate assumption OH = 4 - F is only valid for
elbaitic tourmalines with FeO + MnO < 8 wt%.
Finally,
the conditions
of formation for tourmaline-bearing cavities in the
Zoned Tourmaline from the Himalaya Mine, near Mesa Grande, California